QAD EQMS
4 Reviews 3/5 ★ ★ ★ ★ ★A software solution designed for quality and compliance management in manufacturing. Formerly CEBOS MQ1.
Product Overview
QAD EQMS, formerly MQ1, is a software solution tailored for quality and compliance management in manufacturing settings. It includes document control, compliance oversight, corrective and preventive actions (CAPA), and risk assessment. Additionally, the software facilitates regulatory reporting, audit coordination, and supplier evaluations.
It’s widely adopted by manufacturing firms in various sectors, including life sciences and high-tech. QAD EQMS helps enhance product quality while reducing the costs linked to defective products.
Pros
- Responsive customer service
- Quick record creation and retrieval through documents module
- Can be tailored to specific industries
Cons
- CAPA module could be more configureable
- Limited global search functions
Target Market
Medium to large organizations that need to manage complex quality and compliance requirements in manufacturing, automotive, healthcare, and pharmaceuticals.Not Recommended For
Small businesses requiring more basic QMS functions, or companies outside of the manufacturing industry.QAD EQMS (enterprise quality management system) is an integrated solution designed to manage and improve quality across an entire enterprise. The platform helps streamline manufacturing performance, comply with industry-specific quality standards, and facilitate risk management analysis.
QAD EQMS Key Features
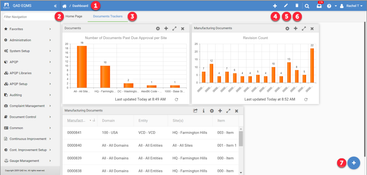
- Document Control: QAD EQMS assists in tracking document revisions and engineering changes, ensuring teams work with the latest document versions. It manages document distribution, changes, and reviews, and lets businesses share them with suppliers.
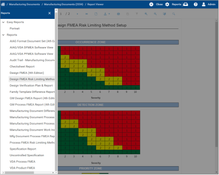
- Quality Management: The system covers continuous improvement and quality management in all parts of an enterprise. This includes modules for managing suppliers, audits, employee performance, risk, various programs, products, PPAP (production part approval process), and APQP (advanced product quality planning) design management.
- Industry-Specific Tools: QAD EQMS is noted for its high level of integration by offering nearly all quality management options. These encompass defect tracking, risk management, and document coding and control, making it useful in industries like healthcare and biotechnology.
- Continuous Improvement Module: The system includes tools for continuous improvement, with features for managing suggestions, projects, lessons learned, issue management, and meetings.
- Corrective and Preventive Actions (CAPA): EQMS identifies issues, implements corrective actions, and tracks their effectiveness to prevent the recurrence of problems.
- Supplier Management: The software manages the quality of products and services provided by suppliers, including supplier evaluations, performance monitoring, and communication.
- Incident Investigation: Allows you to thoroughly investigate non-conformances within the NCR & CAPA module.
Supported Industries
QAD supports several manufacturing-focused industries with varying degrees of regulation.
- Automotive: Compliance with IATF 16949, APQP, and PPAP
- Life Sciences: FDA and ISO compliance, CAPA management
- Consumer Products: Product quality tracking and supply chain audits
- Food and Beverage: FDA and FSMA compliance, traceability, and recall readiness
Product Overview
Developer Overview
Related Products
User Reviews of QAD EQMS
Write a Review- Aerospace & Defense
- 1K-5K employees
I use this software daily, and it's easy to use
We have an older version of CEBOS. I use it daily and have no issues with the software.
Pros
I use this software daily, and it's easy to use. It's like any other software. You have to use it to know the functionality.
Clunky, Buggy, Not User Friendly
Clunky, Buggy, Not User Friendly, Worst Search Functions I’ve Ever Seen… I can’t think of anything positive to say about it.
Pros
when I don't have to use it.
Cons
That one time it renamed hundreds of draft documents to the same title.
- Internet Software & Services
- 11-50 employees
- Annual revenue $1M-$10M
QAD EQMS Review
Antiquated software but uses nice vintage icons
Pros
Vintage Icons
Cons
Yellow on white color combo
- Automotive
- 51-250 employees
- Annual revenue $10M-$50M
QAD EQMS Review
I’ve used the MQ1 solution at my previous job and loved it. I know it has the functionality that I need and know it will work because it’s a great solution