Veeva Vault QMS
A quality management system designed to manage deviations, CAPA, audits, and compliance workflows for life sciences companies.
Product Overview
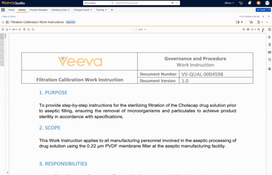
Veeva Vault QMS is a cloud-based quality management system designed for regulated life sciences organizations. It centralizes processes such as deviations, CAPA, audits, change control, complaints, and supplier quality into a single platform.
The system connects quality workflows with documents, training, and risk management so teams can track investigations, enforce compliance requirements, and maintain audit-ready records.
Pros
- Strong compliance with FDA & global regulations
- Integrates with other Veeva tools
- Structured workflows for CAPA, deviations, investigations
Cons
- Expensive for smaller companies
- Implementation can be complex
- Interface can feel complicated
Target Market
Mid-market to enterprise life sciences companies needing a unified compliance QMS.Not Recommended For
Small companies seeking lightweight or low-cost QMS software.Veeva Vault QMS is part of the broader Veeva Vault Quality Suite, which also includes applications for:
- Document control
- Training management
- Validation management
- Quality analytics
Because the applications share the same underlying platform, organizations can connect everything from quality records to supplier activities without moving data between separate systems.
The system is built specifically for pharmaceutical, biotech, and medical device companies. Many workflows reflect common regulatory processes such as deviation investigations, complaint handling, and audit readiness.
Key Features
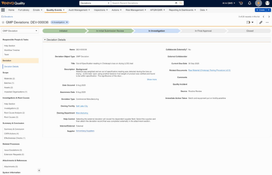
Quality Event Management
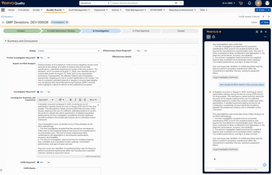
- Handles the full lifecycle of quality events, including deviations, nonconformances, complaints, and audit findings.
- Lets users record events, assign investigations, and track corrective and preventive actions within a single workflow.
- Provides built-in recurrence and duplicate detection to help identify repeated issues before they escalate.
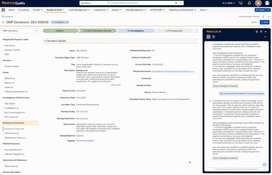
CAPA and Investigation Workflows
- Supports structured root cause analysis and CAPA management with configurable review steps, approvals, and task tracking.
- Links investigations to deviations, complaints, or audit observations for traceable documentation that simplifies inspections.
Audit Management
- Lets users plan, execute, and track internal and supplier audits
- Findings can auto-generate follow-up actions that link to the audit record for compliance documentation.
Change Control
- Allows users to control changes to procedures, equipment, or processes.
- Triggers impact assessments and document revision through change requests so SOPs and training updates remain aligned with operational changes.
Risk Management
- Allows organizations to evaluate and prioritize quality risks across products, suppliers, and manufacturing processes.
- Connects risk records to investigations and CAPA actions to identify patterns and reduce recurring compliance issues.
Supplier Quality Management
- Includes tools for monitoring supplier performance and managing investigations or audit findings.
- Grants external partners controlled access to participate in corrective actions or provide documentation.
Quality Reporting and Dashboards
- Provides built-in dashboards for visibility into trends, including recurring deviations, CAPA timelines, and audit outcomes.
- Helps monitor compliance performance and identify areas where quality processes may be drifting.
Pricing Model
Veeva Vault QMS is sold as a cloud subscription. Pricing is typically customized based on:
- User count
- Modules
- Implementation scope
Large enterprise deployments can include additional Vault Quality Suite applications.
Because the platform is designed for highly regulated industries and large organizations, total costs for enterprise deployments commonly exceed six figures over a multi-year period.
Product Overview
Developer Overview
User Reviews of Veeva Vault QMS
No reviews have been submitted. Do you use Veeva Vault QMS? Have you considered it as part of your software evaluation process? Share your perspective by writing a review, and help other organizations like yours make smarter, more informed software selection decisions!