The Best QMS Software for Medical Devices
With the FDA’s QMSR now aligning 21 CFR Part 820 with ISO 13485, choosing the right QMS has never been more critical for medical device companies. We reviewed the top platforms for design controls, CAPA, regulatory compliance, and audit readiness.
- Responsive customer support
- Intuitive, user-friendly interface
- Automated document routing and revision control
- Completes validation quickly
- Includes automated notification system
- Seamless integration between modules
- Flexible deployment options with access on all devices.
- Has 25+ pre-installed modules for advanced functionality.
- Full validation included for life sciences companies.
Medical device QMS software goes beyond general quality management. They must support design controls, risk management per ISO 14971, and traceability throughout the device lifecycle.
- Greenlight Guru: Best for Medical Device Startups
- MasterControl: Best for Enterprise Compliance
- QT9 QMS: Best All-in-One Mid Market System
- Octave Reliance: Most Configurable
- Qualio: Best for Product Development
- uniPoint: Best for Risk Management
- ComplianceQuest: Best Salesforce-Native QMS
- Unifize: Best Collaboration Tools
Greenlight Guru - Best for Medical Device Startups
Greenlight Guru was purpose-built for medical device manufacturers, offering strong design controls for startups. They function as a true interconnected database, rather than just document storage. This eliminates data silos, allowing you to create and upload ISO 14971-compliant traceability matrices for efficient review. By tracking, documenting, and tracing all design aspects, your team can incorporate risk management into the design process to bring safe products to market faster.
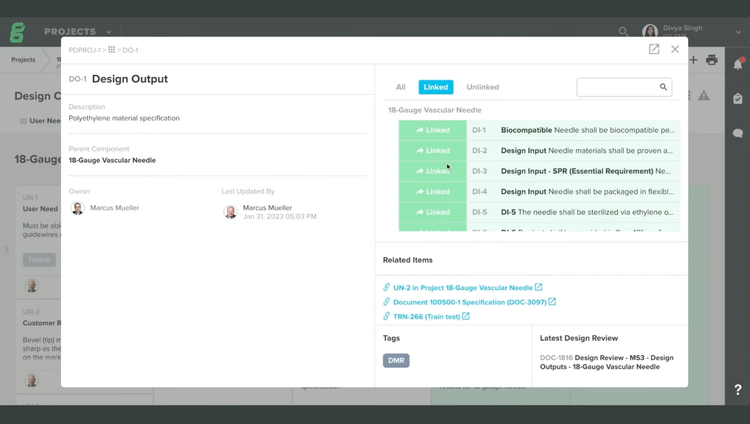
For medical device manufacturers, design controls are one of the most scrutinized areas in FDA submissions and audits. Greenlight Guru helps you comply by linking traceability across user needs, design inputs, verification, and risk on one platform. This way, your team isn’t working around disconnected Excel files and manually reconciling data.
Even better, the system comes preloaded with updated fields and templates that reflect QMSR language, making it easier to comply with the new regulatory requirements. That said, it was originally built for the old QSR terminology and guidelines, so it’s still transitioning, and some workflows don’t always map neatly.
Greenlight Guru is best for small to midsize medical device startups looking for their first full QMS. It has a good balance of strong features without the overhead or high cost and complexity of an enterprise system. Pricing starts at around $600/user/month, though you’ll need a custom quote for exact costs.
MasterControl - Best for Enterprise Compliance
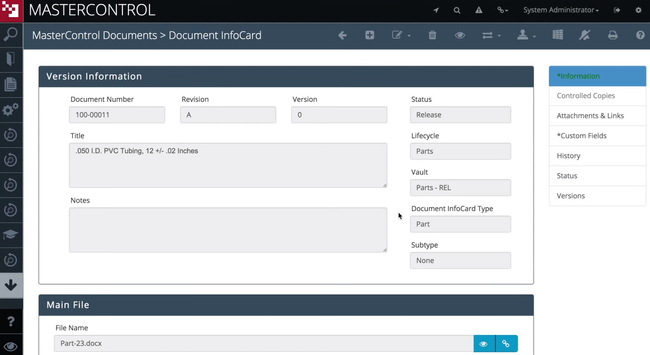
MasterControl is built to support the highly regulated medical device manufacturing industry at scale. The system automates critical processes like document control, CAPA, and change control in a single platform. Even better, the electronic device history record (eDHR) tool captures live data straight from the shop floor. It flags deviations and links them to batch records instantly. This helps you comply with standards like QMSR, ISO 13485, and other global regulatory frameworks.
Additionally, MasterControl offers an advantage for large manufacturers through its connected quality platform. This links quality management with manufacturing, supplier management, and post-market monitoring. This integration improves visibility across the entire product lifecycle, making it easier for large companies to coordinate quality efforts in every department. It also integrates with leading ERP, PLM, and laboratory systems for two-way data sync. This allows you to bring new devices to market faster while maintaining strict regulatory oversight.
That said, because it’s an enterprise-level system, it does have a complex implementation and setup. It often takes months to set up and requires extensive planning, configuration, and training. But if you’re operating a national or global supply chain, this is expected due to the complexity of operations.
QT9 QMS - Best All-in-One Mid Market System
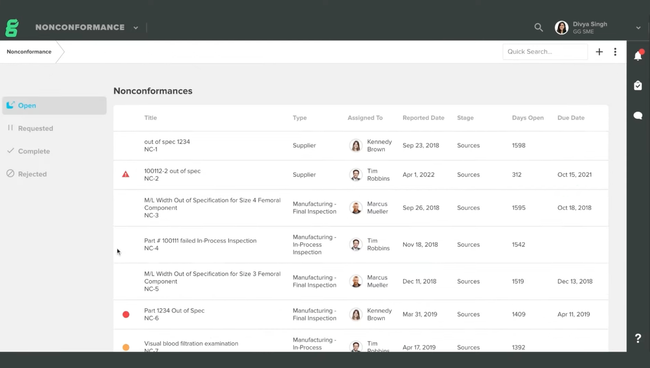
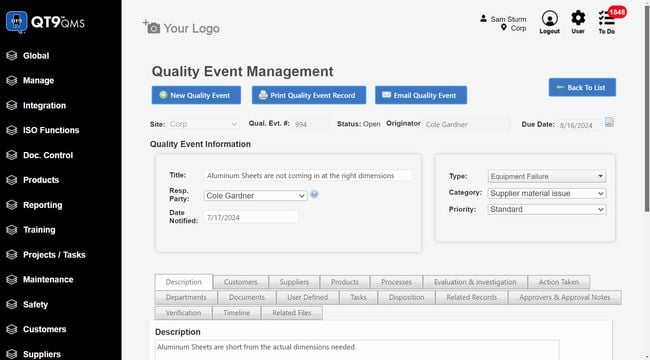
QT9 QMS offers 25+ pre-built modules that cover the full device lifecycle. One module that really stands out is the supplier evaluations. This is crucial in the new FDA QMSR and ISO 13485 regulations. You can monitor supplier performance through structured assessments to ensure they meet your quality, compliance, and delivery standards. It’s a great way to manage risk and improve supplier relationships.
But what makes QT9 a true all-in-one system is its integrated ERP. They share a single database, allowing you to connect quality directly with manufacturing and business operations. You can link quality events with production data, inventory, and purchasing workflows in real time. If you discover a quality issue with a component, manufacturers can quickly trace affected production orders without switching systems.
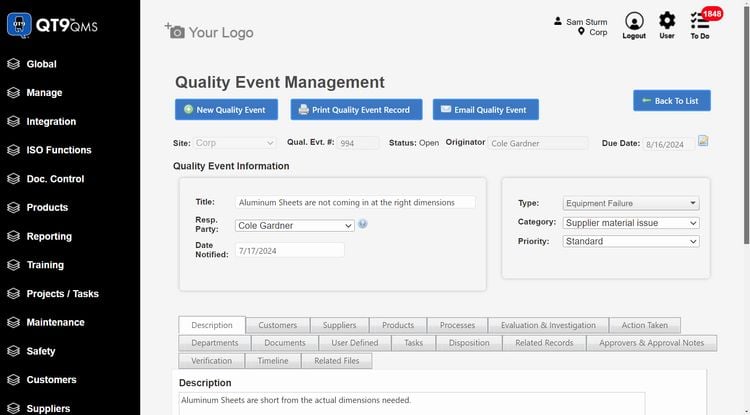
Another advantage of QT9 is that all the QMS modules are included as a single suite. This makes it easier to implement and set up for smaller businesses, as they don’t have to worry about integrating extra modules in the future. It’s a great option for midmarket companies that need ERP + QMS without the enterprise overhead.
Octave Reliance - Most Configurable
Octave Reliance offers a highly configurable closed-loop quality process for manufacturers with diverse product lines. It has over 40 applications, all of which are adaptable to match your organization’s unique workflows, data structures, and reporting needs. This allows you to adjust forms, fields, and structures to your processes, rather than having to change your operations to match the system.
The flexibility is a great advantage for medical device manufacturers, especially those operating across multiple facilities, product lines, and regulatory jurisdictions. For example, you can use no-code tools to build custom CAPA workflows based on issue severity. This way, minor and major nonconformities are automatically routed to different teams and undergo different review processes. You can easily change any workflow when regulations change or when adding new product lifecycles.
Octave Reliance offers an entire QMS suite, including modules for document control, supplier quality, and audits. It’s best for larger companies that require enterprise-level quality management and need integrations with ERP, PLM, and/or MES systems.
Qualio - Best for Product Development
Qualio was built specifically for medical device manufacturing and offers great product development tools combined with a QMS suite. The design control management module lets you link risk assessments, training records, and quality events into a single traceable system. This allows both your quality and product teams to maintain visibility from development and testing to regulatory submissions. Plus, having this level of traceability helps you maintain audit readiness.
Additionally, Qualio has embedded FMEA and ISO 14971 risk management within the design control module. While no software is “ISO certified,” it has built-in tools that make compliance much easier. These include risk matrices, traceability for hazards, and automated risk management file (RMF) generation.
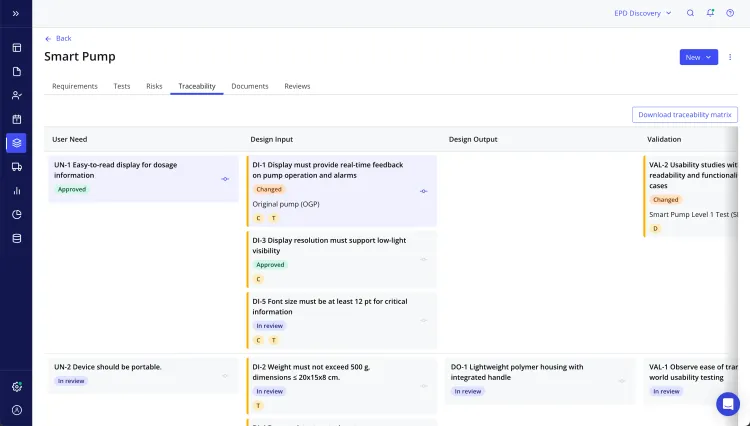
On top of the strong features, Qualio offers an easy-to-use interface that makes navigation simple. The modern design simplifies the layout and helps your team locate documents and quality events. And even better, the clean workflows let your team spend less time training compared to a more complex system.
Qualio is best for growing small to midsize companies looking for their first QMS software. Pricing requires a custom quote, but they offer three different packages that scale in features and users allowed. And while it integrates with CRM and product development systems, it doesn’t have pre-built integrations with any ERPs. This makes it more difficult, as you’ll need to build your own custom integrations via an API or a third-party.
uniPoint - Best for Risk Management
uniPoint includes a strong risk assessment module that gives medical device manufacturers a flexible framework for identifying, scoring, and tracking risks across their production. You can define and manage different types of assessments, like for vendors or parts, with full control over criteria and scoring. Ane because it integrates with the corrective action system, high-risk items can trigger CAPAs without manual handoff.
The module also helps you get audit-ready using color-coded risk levels and symbols, so anyone in the organization can identify and act on high-priority risks. You can also use the built-in reports and metrics to spot risk trends and assess processes without third-party tools. And because it’s so flexible, you can configure your own risk controls to meet your exact needs and shape them to fit ISO 14971 workflows.
uniPoint is best for growing midsize companies that need a configurable QMS suite. While you’ll need a custom quote for your business, we estimate that pricing starts around $225/user/month. Still, it’s highly variable, as it uses a concurrent seat and module-based pricing model.
ComplianceQuest - Best Salesforce-Native QMS
ComplianceQuest is built natively on the Salesforce platform, making it a great option if you already use that CRM. It features a strong closed-loop architecture, helping you centralize your processes from NCs, CAPAs, risk, and document control. The system design also makes it much easier to comply with FDA and ISO 13485 audits. It serves as a comprehensive middle-office application that connects your ERP and CRM.
As for the features, ComplianceQuest provides AI- and machine-learning-powered analytics that highlight patterns across quality events. It can analyze risk trends and perform predictive analytics, enabling you to be proactive in your quality strategy rather than reactive. So whether you’re trying to detect nonconformance or monitoring high-risk issues, the AI can help you catch them before they become recalls.
That said, you’ll need someone with strong Salesforce administration expertise to properly run ComplianceQuest. So if you don’t have someone with this experience, or don’t have Salesforce at all, this probably is not for you. But if you do, it’s best for large med device companies with over 500 employees.
Unifize - Best Collaboration Tools
Unifize takes a different approach to quality management by embedding real-time conversations directly into quality records. So every CAPA, nonconformance, or document review carries its one threaded conversation. This way, the context behind the decisions lives alongside the record, rather than scattered across Slack or email inboxes. And for medical device teams who work across departments like engineering and quality, this eliminates a huge efficiency bottleneck.
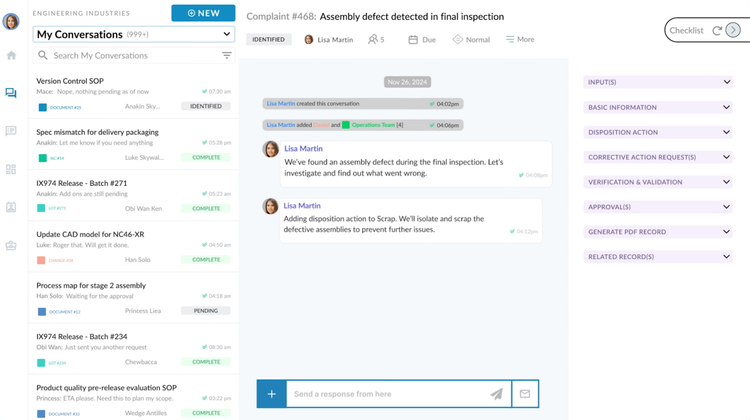
Additionally, the strong collaboration really stands out in its easy-to-use document management system. Here, you can draft an SOP, and the system can embed conversations directly into the document record. So the review discussion, history, and approval workflow all live in the same place. It has a no-code builder that lets you build workflows by just dragging and dropping processes. Important documents are routed to the correct people, and any changes or approvals are noted right in the system for greater efficiency.
Unifize also has public pricing, which is rare in the quality management software market. For the QMS and document management system (DMS), it starts at $2,340/org/month with up to 20 full users. It’s a great option for growing mid-sized to enterprise companies looking for an all-in-one QMS.
What is Medical Device QMS Software
A medical device quality management system (QMS) is a platform that manages the quality processes required to design, manufacture, and maintain medical devices under standards like ISO 13485 and FDA 21 CFR Part 820 (now FDA QMSR). Many times, these are integrated with medical device manufacturing ERPs to connect production and quality data.
Unlike general-purpose quality management systems that handle document control and CAPAs across any industry, a medical device QMS is built around the specific workflows regulators expect: design controls, risk management traceability per ISO 14971, device history records, and complaint handling tied to post-market surveillance.
How QMSR Changes What You Need From a QMS
As of February 2, 2026, the FDA’s Quality Management System Regulation (QMSR) formally replaces the old 21 CFR Part 820 framework with one built around ISO 13485. This changed the structure of what the FDA expects to see in your QMS. Companies that built their QMS around the old Part 820 language now need to ensure their processes, documentation, and terminology align with ISO 13485’s process-based approach. Here are some things to look out for when evaluating software for QMSR compliance:
- Risk Management Built-In: Risk decisions must link to design, purchasing, production, and CAPA workflows; not siloed in a standalone module.
- Traceability: Traceability needs to be across the entire system and fully auditable.
- Supplier Quality Management: Audit scheduling and performance tracking for each supplier are key now that they’re inspectable.
Further Reading: FDA QMSR Explained: What Changed and What It Means for Your QMS
Key Regulations and Standards for Medical Device Companies
Medical device manufacturers must comply with several regulations and standards, including:
| Regulation / Standard | What It Governs | Priority |
|---|---|---|
| ISO 13485:2016 | International QMS standard for medical devices. Covers the full product lifecycle, including design, manufacturing, distribution, and servicing. Required or effectively required for market access in the EU, Canada, Australia, and most global markets. Now incorporated into the FDA’s QMSR. | Essential |
| FDA QMSR (formerly 21 CFR Part 820) | U.S. FDA Quality Management System Regulation for medical device manufacturers. As of February 2026, the updated QMSR formally aligns Part 820 with ISO 13485. Applies to any finished device manufacturer selling in the U.S. | Essential |
| FDA 21 CFR Part 11 | Governs electronic records and electronic signatures. Sets criteria for when digital records and e-signatures are considered trustworthy and equivalent to paper. Non-negotiable for any QMS used in FDA-regulated environments. | Essential |
| ISO 14971:2019 | International standard for risk management applied to medical devices. Covers hazard identification, risk estimation, risk control, and residual risk evaluation across the entire product lifecycle. Referenced by both ISO 13485 and the FDA. | Essential |
| EU MDR (2017/745) | European Medical Device Regulation. Governs safety, performance, and post-market surveillance of medical devices sold in the EU. Requires technical documentation, clinical evaluation, and UDI compliance. This is essential if you place applicable devices on the EU market. | Essential (EU) |
| EU IVDR (2017/746) | European In Vitro Diagnostic Regulation. Similar framework to EU MDR, but specific to diagnostic devices. Requires stricter classification, performance evaluation, and post-market follow-up for IVDs sold in the EU. | Situational |
| MDSAP | Medical Device Single Audit Program. Allows a single audit to satisfy requirements across multiple jurisdictions (U.S., Canada, Australia, Brazil, Japan). Uses ISO 13485 as its core framework. Mandatory in Canada; voluntary elsewhere. | Recommended |
| IEC 62304 | International standard for the software lifecycle of medical device software. Defines requirements for development, maintenance, risk management, and configuration management of software used in or as a medical device. | Situational |
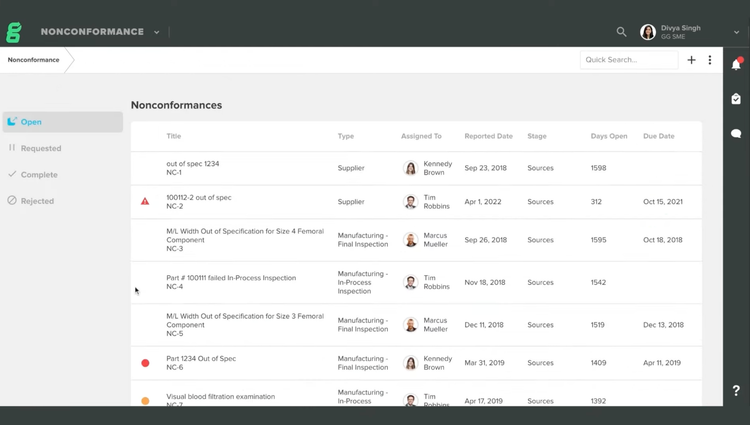
Key Features
Here are the main functionalities of medical device QMS software:
| Key Features | Benefits |
|---|---|
| Document Control & Version Management | Ensures every team member works from the current approved version. Creates the audit trail regulators expect for controlled documents. |
| Design Controls (DHF, DMR, DHR) | Structures the entire design process from user needs through verification and validation. Produces the design history file for the FDA and notified bodies’ review during audits. |
| CAPA Management | Systematically captures, investigates, and resolves quality issues. Demonstrates to regulators that your organization identifies root causes and prevents recurrence. |
| Risk Management (ISO 14971 / FMEA) | Links hazard identification, risk estimation, and risk controls to design and production records. Required by both ISO 13485 and FDA for the entire product lifecycle. |
| Training Management | Tracks employee training assignments, completions, and competency records. Proves to auditors that personnel are qualified to perform their roles. |
| Audit Management & Audit Trails | Plans, schedules, and documents internal and external audits in one system. Provides the timestamped, tamper-evident records required under 21 CFR Part 11. |
| Supplier Quality Management | Monitors supplier performance, qualifications, and corrective actions. Keeps incoming material risk visible and traceable back to specific suppliers. |
| Change Management | Controls how changes to designs, processes, or documents are proposed, reviewed, and approved. Prevents unapproved changes from reaching production or the field. |
| Electronic Signatures (21 CFR Part 11) | Replaces wet signatures with compliant e-signatures tied to user identity and intent. Non-negotiable for any QMS used in an FDA-regulated environment. |
| Complaint Handling & Post-Market Surveillance | Captures customer complaints and field issues in a structured workflow. Feeds post-market data back into risk management and CAPA processes, as required by regulators. |
| Nonconformance Management | Documents and dispositions materials, components, or products that don’t meet specifications. Creates traceability between nonconforming items, root cause investigations, and corrective actions. |
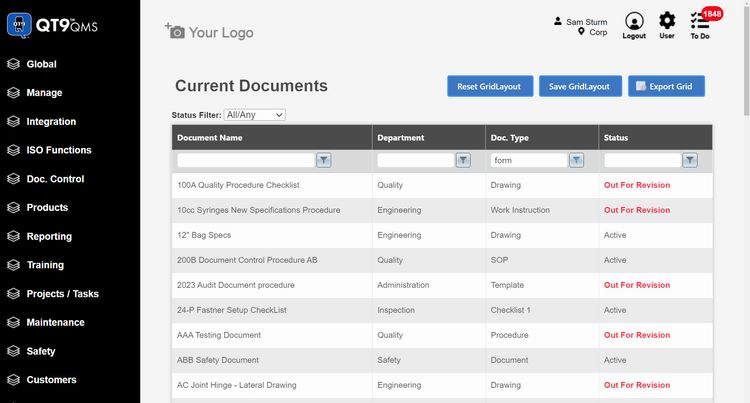
Pricing Guide
Medical device QMS software can range anywhere from $5,000 to over $150,000 per year. The pricing is extremely variable because it’s based on a number of different factors:
- Total user count
- Required modules
- Regulatory scope
- Process complexity
- Data migration or required customizations
Here is a general breakdown of market segments and what you can expect to pay based on your business size. Note that these are approximate and can vary depending on your specific implementation.
| Tier | Company Size | Total Cost of Ownership (TCO) | Example Software |
|---|---|---|---|
| Low-Tier | 1–25 employees | $5,000–$15,000 per year | Qualio, QT9 QMS |
| Mid-Tier | 25–150 employees | $20,000–$50,000 per year | Greenlight Guru, Intellect QMS |
| High-Tier | 150–500 employees | $50,000–$150,000 per year | MasterControl, Octave Reliance |
| Enterprise | 500+ employees | $150,000+ per year | Veeva Vault QMS, ComplianceQuest |