What Is ICH Q10?
ICH Q10 defines how pharmaceutical companies should manage quality across the entire product lifecycle, from development through discontinuation.
A lot of it overlaps with existing GMP rules, and technically, only the parts that go beyond GMP are optional. But in reality, regulators in the US, EU, and Japan still expect companies to follow it closely. The right QMS software makes it easier to manage processes, maintain documentation, and stay audit-ready without adding complexity.
What Is ICH Q10?
ICH Q10 is a guideline published in 2008 by the International Council for Harmonisation (ICH). ICH is a group that brings together regulators and pharmaceutical companies across the US, EU, Japan, and other regions. At its core, it explains how to run a pharmaceutical quality system across the entire product lifecycle.
ICH Q10 builds on ISO 9001, the global standard for quality management systems. However, it is specifically adapted for drug manufacturing. It incorporates GMP requirements, adds a lifecycle focus that ISO 9001 doesn’t cover, and focuses on risk-based decision-making.
Much of what ICH Q10 outlines is already covered by regional GMP regulations. So it’s not entirely optional. The parts that align with GMP are effectively required through those regulations. The “optional” pieces are those that go beyond GMP and mostly apply to development-stage activities or areas where ICH Q10 raises the bar beyond minimum compliance.
The Regulatory Landscape
ICH Q10 is recognized across major pharmaceutical regulatory regions.
- US: The FDA published it as official guidance in 2009 and noted that much of it aligns with existing requirements under 21 CFR Parts 210 and 211.
- EU: Included as Part III of the EU GMP Guide; the European Medicines Agency evaluates its principles during GMP inspections, with similar recognition from organizations like the WHO and MHRA.
ICH Q10 isn’t meant to replace or expand GMP requirements. Instead, it builds on them. The parts that overlap with GMP are effectively required. Anything that goes beyond GMP is technically optional. In practice, though, regulators expect companies to show the kind of structured, lifecycle-focused quality approach that ICH Q10 describes.
It’s also separate from the FDA’s Quality Management Maturity (QMM) program. QMM is a voluntary initiative from the FDA’s Center for Drug Evaluation and Research (CDER) that shares some of the same ideas.
However, QMM assessments are not used to determine cGMP compliance. Instead, they focus on how a company actually operates: things like behaviors, processes, and overall quality culture. The goal is to encourage manufacturers to go beyond minimum GMP requirements. Companies that already align with ICH Q10 principles are generally better positioned for it, but the two should still be treated as distinct.
ICH Q8, Q9, and Q10: The Tripartite Framework
ICH Q10 is part of a broader set of guidelines that work together to shape how pharmaceutical companies manage quality:
| Guideline | Focus | Description |
|---|---|---|
| ICH Q8 | Pharmaceutical Development | Designs drugs and processes using quality by design (QbD) to build quality in upfront. |
| ICH Q9 | Quality Risk Management | Identifies, assesses, and controls risks to product quality across the lifecycle. |
| ICH Q10 | Pharmaceutical Quality System | Connects development and risk practices into a system used in daily operations. |
There’s no required order for implementing these, and they’re meant to complement each other. But in practice, Q10 works best when it’s backed by Q8 and Q9. Together, they form what ICH calls a “tripartite” approach to pharmaceutical quality. Two core enablers sit underneath all three, connecting high-level guidance in Q8, Q9, and Q10 to daily decisions on the floor:
- Knowledge management ensures that what you learn during development, tech transfer, manufacturing, and post-market is actually captured and reused.
- Quality risk management (QRM) keeps decisions grounded in risk. It feeds into changes, investigations, and ongoing process improvements.
Objectives and the 4 Core PQS Elements
Three Core Objectives
ICH Q10 defines three core objectives that describe what a pharmaceutical quality system (PQS) should deliver:
1. Product realization: Ensure products consistently meet performance, patient, and regulatory requirements across the full lifecycle. 2. State of control: Keep processes stable, monitored, and predictable, so quality issues are prevented in the first place. 3. Continual improvement: Build ongoing improvement into operations, not just after problems occur.
These objectives directly shape how the PQS is structured and evaluated.
The 4 Core PQS Elements
ICH Q10 breaks the PQS into four operational areas where companies need clear, well-documented systems, often supported by eQMS software:
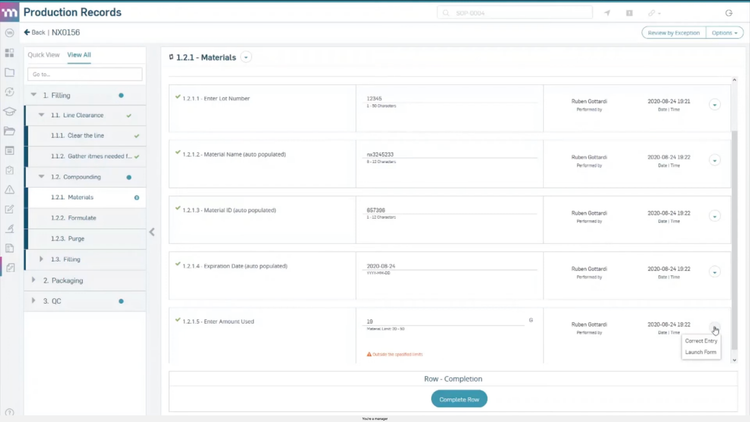
1. Process Performance and Product Quality Monitoring System: Tracks how processes and products perform using quality data and trend analysis. The goal is to maintain control and catch issues early, before they impact quality.
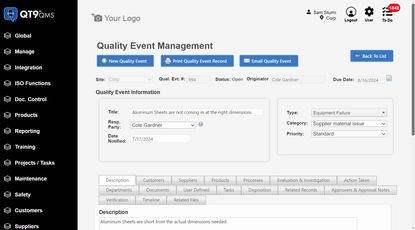
2. Corrective and Preventive Action (CAPA) System: Manages how issues are investigated and resolved. This includes identifying root causes, applying corrective actions, and preventing recurrence. Strong documentation is critical to ensure traceability and audit readiness.
3. Change Management System: Controls how changes are evaluated, approved, and implemented. This ensures updates to processes, materials, or equipment don’t introduce new risks and creates a clear record of what changed and why.
4. Management Review: Gives leadership visibility into PQS performance through metrics, trends, CAPA activity, and changes. It connects quality data to decisions around risk, resources, and improvement priorities.
Lifecycle and Management Responsibilities
The Four Lifecycle Stages
ICH Q10 applies across the full product lifecycle, with different priorities at each stage but one continuous PQS:
| Lifecycle Stage | Description |
|---|---|
| Pharmaceutical Development | Builds product and process understanding; adds structure where GMP is limited. |
| Technology Transfer | Transfers knowledge to manufacturing or new sites without losing critical details. |
| Commercial Manufacturing | Maintains process control and product quality under full GMP requirements. |
| Product Discontinuation | Manages product withdrawal, record retention, and ongoing compliance obligations. |
Management Responsibilities
Across all stages, ICH Q10 requires active management involvement. Leadership is expected to:
- Set and communicate quality policy
- Allocate resources to support the PQS
- Review system performance regularly
This also extends to outsourced manufacturing and suppliers. Even when production is contracted out, the sponsor remains responsible for product quality and compliance.
ICH Q10 Compliance
Implementing ICH Q10 involves an overall shift in how quality is managed across the organization, rather than a one-time rollout. Most companies approach it in phases:
Gap assessment Start by comparing your current quality system to ICH Q10 PQS elements. Review how CAPA, change management, and management reviews actually function to identify gaps between current practices and ICH Q10 expectations.
Management commitment ICH Q10 depends on active leadership involvement. That includes executive sponsorship, clear quality policies, defined roles, and real investment in quality systems.
Define roles and build out PQS elements Each PQS element needs ownership, documented SOPs, and consistent execution. This typically involves updating procedures, training teams, and setting measurable KPIs to track performance and improvement.
Common Challenges
Paper-based systems Manual or fragmented systems struggle to support ICH Q10. They lack reliable audit trails and make real-time monitoring and trend analysis difficult.
Multi-site consistency Maintaining aligned PQS processes across multiple sites, including outsourced manufacturing, is complex. ICH Q10 expects consistent standards, which requires active oversight and governance.
Quality culture vs. compliance It’s easier to track compliance outputs than to measure and improve quality culture. ICH Q10 pushes beyond documentation into how quality is actually managed day to day.
The Role of eQMS Software
Paper-based systems and tools like shared drives, spreadsheets, and email don’t hold up under ICH Q10. As expectations around data integrity and quality system maturity increase, gaps in process controls and audit trails become unmanageable.
An electronic quality management system (eQMS) turns PQS requirements into structured, repeatable workflows aligned directly to the four PQS elements.
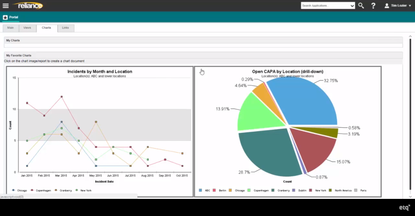
- Process Performance and Product Quality Monitoring: Provides real-time dashboards and quality metrics; helps teams quickly analyze trends and investigate issues across products, processes, and sites.
- CAPA: Automates CAPA workflows from deviations, complaints, or audits. Supports root cause analysis, assigns actions with ownership and deadlines, and verifies effectiveness.
- Change Management: Controls how changes are requested, risk-assessed, approved, and implemented; links changes to training, documentation, and process updates with full traceability.
- Management Review: Centralizes KPIs and performance data into audit-ready reports; gives leadership a clear, current view of PQS performance without gathering data manually.
Electronic Records Compliance: 21 CFR Part 11 and EU Annex 11
An eQMS must also meet electronic records requirements:
- FDA 21 CFR Part 11: Defines how electronic records and signatures are considered trustworthy and equivalent to paper.
- EU Annex 11: Governs computerized systems used in GMP-regulated environments.
Both require system validation, secure audit trails, role-based access, electronic signatures, and strong data integrity controls. These requirements directly affect how an eQMS handles CAPA records, change approvals, deviations, and management review data. If a system supports ICH Q10 workflows but doesn’t meet 21 CFR Part 11 or EU Annex 11, it creates a separate compliance risk.
Top QMS Software
Frequently Asked Questions
Is ICH Q10 mandatory?
What’s the difference between ICH Q10 and GMP?
What are the 4 elements of ICH Q10?
The four PQS elements are:
- Process Performance and Product Quality Monitoring
- CAPA (Corrective and Preventive Action)
- Change Management
- Management Review
Together, they support ICH Q10’s core objectives: product realization, state of control, and continual improvement.
