Best Pharmaceutical QMS Software
We’ve assessed the top pharmaceutical quality management systems with pros, cons, and pricing. Our top picks cover vital features, including risk management, deviation tracking, and supplier qualification.
- Completes validation quickly
- Includes automated notification system
- Seamless integration between modules
- Strong compliance with FDA & global regulations
- Integrates with other Veeva tools
- Structured workflows for CAPA, deviations, investigations
- Flexible deployment options with access on all devices.
- Has 25+ pre-installed modules for advanced functionality.
- Full validation included for life sciences companies.
Pharmaceutical QMS software handles the hard parts of FDA 21 CFR Part 11 and cGMP compliance: document control, CAPA, batch records, training, and audit management in one validated platform. Here’s how 8 options compare.
- MasterControl: Best for Enterprise Pharmaceuticals
- Veeva Vault QMS: Best for Biopharmaceuticals
- QT9 QMS: Best for Small-to-Mid-Size Manufacturers
- Qualio: Best for Pre-Commercial Pharmaceuticals
- TrackWise: Best for High-Volume Manufacturing
- Octave Reliance: Best for Cross-Regulated Operations
- ComplianceQuest: Best for Salesforce Users
- QAD EQMS: Best Supplier Quality Management Tools
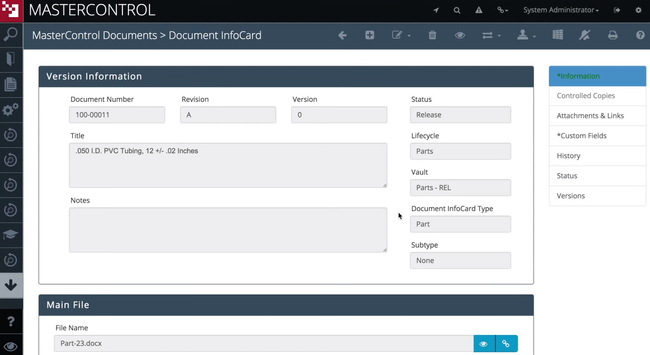
MasterControl - Best for Enterprise Pharmaceuticals
MasterControl gives enterprise manufacturers in-depth quality process coverage, from change and document control to nonconformance handling and risk management. Rather than relying on separate add-ons, it provides module-to-module connectivity: deviations automatically tie to investigations, CAPA, and impacted documents without manual intervention.
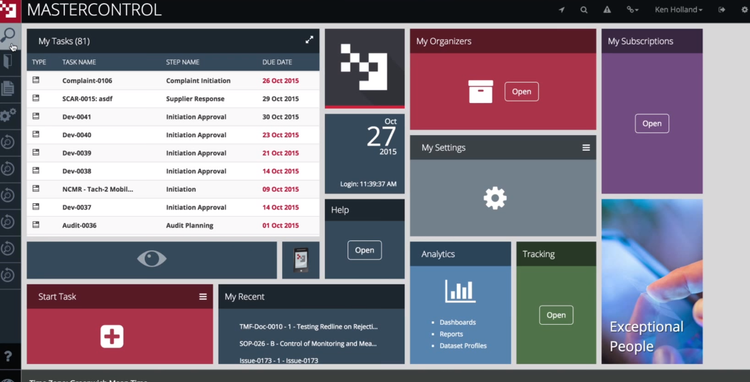
For enterprise pharma teams managing QA, QC, regulatory, and manufacturing workflows, that means fewer gaps between systems and fewer compliance issues. MasterControl also provides data analytics capabilities that uncover trends across quality events. This helps larger companies transition from reactive firefighting to proactive risk identification.
That said, more connected modules and multiple sites mean more validation and re-validation with every software update. For that reason, MasterControl includes a patented Validation Excellence Tool (VxT) that automates much of the GAMP 5 process. Plus, the system includes pre-configured compliance templates for FDA 21 CFR Part 11, 21 CFR Parts 210/211, and ISO standards, which can ease your deployment burden during a lengthy implementation process.
Dive into our full review of MasterControl to learn more.
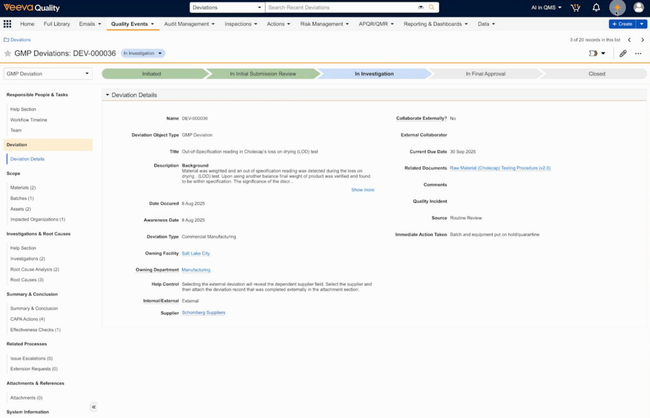
Veeva Vault QMS - Best for Biopharmaceuticals
Veeva Vault QMS tends to fit well for biotech and broader life sciences manufacturers. It’s designed to handle core processes in biopharma development and commercialization, connecting quality to clinical trial data, regulatory submissions, safety, and manufacturing.
As part of Veeva’s Quality Cloud, it lives in the same Vault platform as their safety, regulatory (RIM), and clinical (CTMS) applications. That means a complaint logged in the QMS can flow directly into Safety for adverse event processing. A change control launched in the QMS can auto-trigger a regulatory assessment in RIM.
Veeva also provides cross-functional integration. For example, a deviation at your contract manufacturing site triggers a product-level change, the QMS handles the quality investigation while RIM covers the regulatory impact assessment. If you work with CMOs, contract labs, and suppliers, Veeva also provides them with secure, time-limited access to collaborate on investigations and corrective actions.
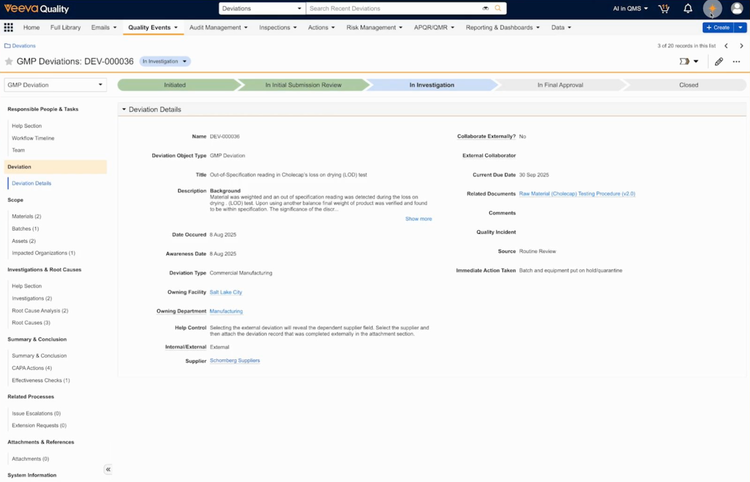
The tradeoff is cost and complexity. Veeva is priced for enterprise pharma, and implementation requires significant planning and internal resources. If you’re a smaller team without a dedicated QA system, you’ll likely find the platform oversized for your needs.
Learn more about Veeva Vault QMS.
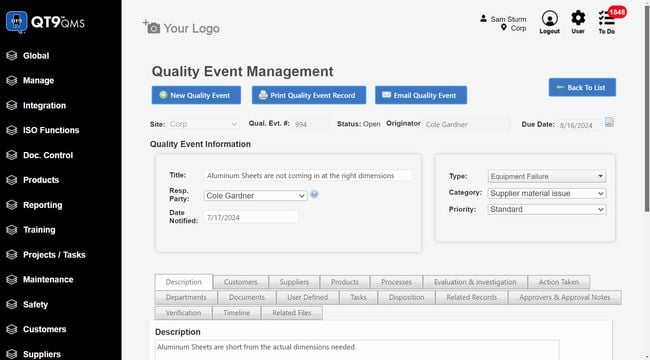
QT9 QMS - Best for Small-to-Mid-Size Manufacturers
QT9 QMS is a solid pick for pharmaceutical manufacturers that need full regulatory coverage without the enterprise price tag. All modules come included in the base subscription, including:
- CAPA
- Document control
- Audit management
- Training
- Risk management
- Change control
- Complaint handling
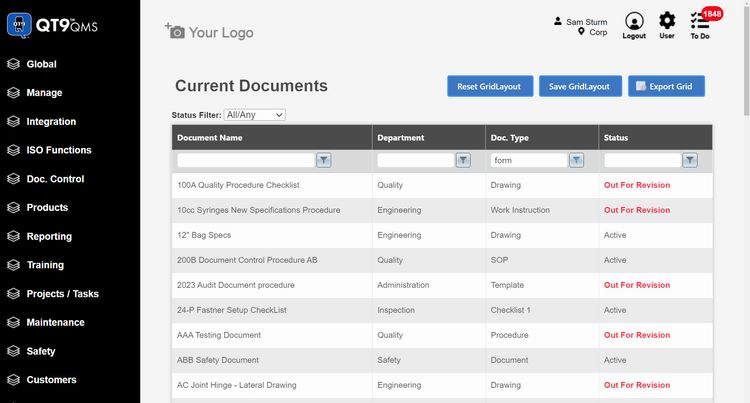
Where QT9 earns its spot for pharma specifically is electronic batch records (EBRs) and master batch records (MBRs). MBRs define your raw material formulas, manufacturing steps, equipment requirements, and quality control checkpoints, all aligned to cGMP standards. EBRs then capture the actual production data for each batch.
QT9 is also pre-validated, with full IQ/OQ/PQ documentation; this is a boon for smaller companies, as it adds no extra cost with every version upgrade. It also supports compliance with FDA 21 CFR Parts 210/211, ICH Q10, PIC/S, ISO 9001, ISO 17025, and EU GMP Annex 11. Plus, concurrent licensing means you’re paying for simultaneous users rather than named seats, which keeps costs down.
QT9’s base package starts at $2,200 per concurrent user per year. Implementation and initial training are a separate one-time fee, but all ongoing training is unlimited and free.
See our review of QT9 QMS for pros, cons, and pricing details.
Qualio - Best for Pre-Commercial Pharmaceuticals
Qualio QMS is ideal for early-stage pharmaceutical companies that need a quality system compliant with FDA and GxP requirements, though it can also support some commercial‑stage firms. Its core strength is document management with built-in collaboration. With its in-app editor, your team can create and approve SOPs, technical documents, and quality records without toggling between different systems.
The conversion tool pulls in legacy files like spreadsheets, legacy records, and paper documents, so you’re not rebuilding your documentation from scratch. Everything lives in a centralized, version-controlled environment with embedded e-signatures that comply with 21 CFR Part 11 and EU GMP Annex 11.
Qualio also includes AI-driven generation of change summaries. This saves your QA team time on routine document updates and gives auditors a clear, consistent change narrative. The tradeoff is that Qualio is designed for companies preparing for regulatory approval rather than full commercial manufacturing scale. Pricing starts at $12,000/year for the platform, plus $3,000 per user per year.
Find out if Qualio QMS is right for your operations.
TrackWise - Best for High-Volume Manufacturing
TrackWise is built for manufacturers generating hundreds of deviations per month across production lines. Its CAPA and deviation management engine enables parent-child record relationships. So when a deviation triggers an investigation and spawns a CAPA requiring change control, each record stays linked with full traceability. If you’re processing dozens of batches daily, this means deviations won’t get lost.
TrackWise also handles:
- Complaint management
- Out-of-specification (OOS) investigations
- Audit management
- Supplier quality. These connect through configurable workflows that reflect your existing SOPs rather than rigid templates.
The platform supports compliance with FDA 21 CFR Part 11, 21 CFR Parts 210/211, EU GMP Annex 11, and GxP standards via robust audit trails and electronic controls.
Tradeoffs include enterprise pricing and implementation complexity. Small-to-mid-sized pharmaceutical teams may find it oversized; however, it’s ideal for high-volume operations.
Learn more in our full TrackWise review.
Octave Reliance - Best for Cross-Regulated Operations
If you’re managing cGMP compliance alongside environmental permits, occupational safety programs, chemical handling protocols, or supplier quality across non-pharma product lines, Octave Reliance is built for that complexity. It provides over 40 configurable applications spanning EHS and enterprise risk management.
For the pharmaceutical industry specifically, Octave offers a Life Sciences Compliance application that:
- Supports FDA 21 CFR Part 11
- Provides validation services
- Enables eMDR-compliant complaint handling
- Supports planned deviation management.
But what makes Octave the pick for cross-regulated operations is what surrounds that pharmaceutical core: supplier corrective action requests (SCARs) via a secure vendor portal, plus enterprise risk tools such as compliance obligations management and emergency preparedness.
Read our full Octave Reliance review for more details.
ComplianceQuest - Best for Salesforce Users
ComplianceQuest is built natively on the Salesforce platform, leveraging Salesforce’s underlying data model, security, and user interface. For pharmaceutical organizations already invested in Salesforce, this native integration simplifies data sharing across departments compared with standalone QMS platforms that require custom integrations.
The platform offers end‑to‑end complaint management with workflows that support adverse‑event reporting and pharmacovigilance‑aligned processes. It captures complaints, links them to investigations and CAPA workflows, and helps track quality events from initial report to resolution.
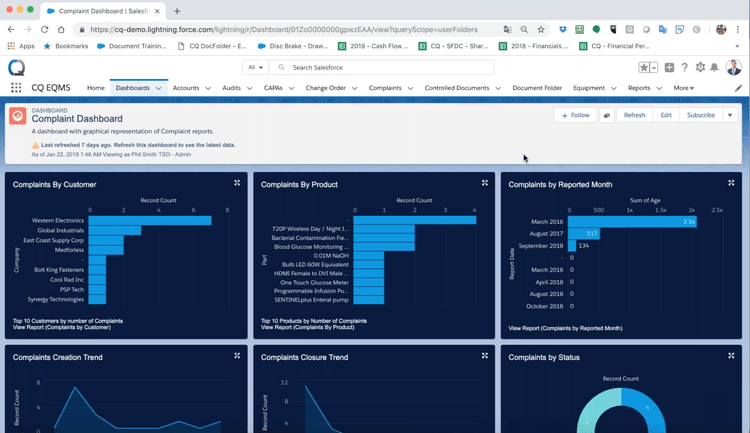
The QMS also interfaces with ERP, MES, LIMS, HR, and PLM systems via APIs and pre‑built connectors, enabling data sharing across quality and operational systems. It also includes analytics that surface patterns across quality events, using AI‑driven insights to help teams shift from reactive resolution to proactive risk identification.
The tradeoff is complexity. Users report a steep learning curve and a complex backend, particularly around user permissions and configuration. Having Salesforce admin expertise is helpful. You can adopt ComplianceQuest without Salesforce, but the platform’s greatest value is unlocked when paired with a Salesforce environment.
Read our full review of ComplianceQuest to learn more.
QAD EQMS - Best Supplier Quality Management Tools
QAD EQMS helps manage the quality of every raw material supplier, contract manufacturer, and packaging vendor in their supply chain. Its supplier quality tools let you evaluate vendor performance and conduct supplier audits with customizable scoring templates.
The system syncs supplier management with:
- Advanced product quality planning (APQP)
- CAPA
- Nonconformance reporting
- Complaint management
In other words, when a defective raw material shipment triggers a nonconformance, the investigation links back to the supplier record, a corrective action is issued, and the resolution is reflected in that supplier’s performance score. Suppliers can then interact directly through self-service portals to implement corrective actions.
QAD also auto-calculates chargeback amounts for defective shipments and automates notifications when supplier certifications (ISO 9001, GMP, or otherwise) are approaching expiration. For pharma companies sourcing excipients and packaging materials from a global supplier base, this proactive oversight helps prevent quality gaps from reaching the production floor.
Learn more about features, pros, and cons in our QAD EQMS review.
What Is Pharmaceutical QMS Software?
Pharmaceutical QMS software is a platform that helps drug manufacturers automate quality processes, documentation, and compliance to ensure medicines are safe and effective.
In the pharmaceutical industry, a quality management system is a documented structure of processes, controls, and responsibilities that governs how a company develops, manufactures, and distributes drugs. ICH Q10 defines this framework as a pharmaceutical quality system or PQS.
General-purpose QMS software covers basic functions such as CAPA, document control, and audit management. However, pharmaceutical QMS supports regulatory needs for this sector:
- FDA 21 CFR Part 11 electronic signature requirements
- Batch record management
- Deviation and OOS investigations
- cGMP workflows under 21 CFR Parts 210/211
- Digital audit trails for EMA and FDA inspectors
Paper-Based QMS vs. eQMS
A paper-based QMS means managing SOPs in binders and tracking training in spreadsheets. Because so many processes remain manual, human error can pose compliance risks.
An eQMS is the digital execution of a quality management system. It integrates all processes into a single, validated platform with role-based permissions, automated workflows, electronic approvals, in-depth audit trails, training management, and version control.
Key Pharmaceutical Regulations
Depending on where you manufacture and sell, pharmaceutical regulations can vary widely, ranging from FDA requirements to international standards and validation frameworks.
| Regulation/Standard | What It Governs |
|---|---|
| FDA 21 CFR Part 11 | Electronic records & electronic signatures |
| FDA 21 CFR Parts 210/211 | cGMP for pharmaceutical manufacturing |
| ICH Q10 | Pharmaceutical Quality System framework |
| GxP | Umbrella term: GMP, GLP, GCP |
| ISPE GAMP 5 | Software validation framework for pharma |
| EU GMP Annex 11 | Computerized systems in EU pharma |
| PIC/S GMP | International pharmaceutical inspection standards |
| ISO 9001:2015 | General quality management system standard |
| GMP / cGMP | Good Manufacturing Practice (umbrella) |
FDA compliance starts with 21 CFR Parts 210 and 211. Part 210 defines Current Good Manufacturing Practices (cGMP) requirements for finished pharmaceuticals, which span equipment maintenance, facility design, production controls, laboratory testing, and recordkeeping.
21 CFR Part 11 builds on this by laying the groundwork that digital QMS must satisfy for electronic records and electronic signatures. This can include tamper-evident audit trails and secure user authentication.
Internationally, ICH Q10 provides the broad framework for a pharmaceutical quality system. GxP covers GMP, Good Laboratory Practice (GLP), and Good Clinical Practice (GCP) that cover everything from research through distribution across the drug lifecycle.
ISPE GAMP 5 is the industry standard framework for software validation in GxP environments. EU GMP Annex 11 controls computerized systems used in pharmaceutical manufacturing within Europe.
Recent Developments
- CSA Replaces CSV (September 2025). After the FDA completed its guidance on Computer Software Assurance, it shifted software validation to a risk-based model focused on functions impacting patient safety, product quality, and data integrity. We recommend asking vendors whether their validation aligns with CSA, so you’re not inheriting an outdated documentation structure.
- 21 CFR 211.110 Draft Guidance (January 2025). FDA requires eQMS infrastructure with live data capture and MES/LIMS integration for advanced pharmaceutical manufacturing operations, like real-time release testing, in-line monitoring, and continuous process verification. Ensure your QMS can process continuous production data.
Key Features
- Audit management: Spans internal and external audit planning, scheduling, and tracking; manages findings and ties them to corrective actions so you’re not scrambling when an inspector arrives.
- Batch record management: Includes electronic batch records (EBRs) that capture execution data per production run and master batch records (MBRs) that define materials, formulas, equipment, and process steps.
- CAPA management: Helps log nonconformances, conduct root cause analysis, deploy corrective actions, check effectiveness, and close the loop with preventive measures.
- Change control: Documents any process, document, equipment, or material change and assesses impact; records approvals and makes records traceable.
- Complaint management: Manages product complaints and adverse event reporting with structured intake, escalation rules, and regulatory reporting workflows.
- Deviation and nonconformance management: Logs failed in-process tests, temperature excursions, missed batch record steps; routes them through investigation and root cause analysis.
- Document control: Includes stringent version control and formal review/approval cycles for SOPs, batch records, specifications, and validation protocols.
- Electronic signatures and audit trails: Secure authentication, time-stamped change records, and tamper-evident logs that capture who did what, when, and why under 21 CFR Part 11.
- Risk management: Provides tools like FMEA, risk registers, scoring matrices, and mitigation tracking to help flag quality risks before they become deviations.
- Supplier management: Helps qualify, onboard, and monitor raw materials suppliers, API manufacturers, and contract labs for cGMP.
- Training management: Links training records to specific procedures; auto-assigns training for revised SOPs and blocks uncertified employees from performing regulated tasks until training is completed.
Pharmaceutical QMS Pricing
Ballpark pharmaceutical QMS pricing figures by company size, from $10,000/year for startups to over $150,000/year for enterprise manufacturers:
Startups / small pharma ($10K–$20K/yr)
- Covers audit management, CAPA, document control, and training management
- Includes Qualio and QT9 QMS at low user counts
- Plan for a one-time implementation fee
Mid-market ($20K–$50K/yr)
- Includes deeper integrations, batch records, complaint handling, and supplier quality management
- Includes QAD EQMS, ComplianceQuest, and QT9 at scale
High-tier ($50K–$150K/yr)
- Involves multi-site operations managing cGMP, EHS, and supplier quality
- Includes MasterControl, TrackWise, EtQ Reliance
- May require consultants due to longer implementations
Enterprise ($150K+/yr)
- Global pharma with cross-functional needs spanning regulatory, clinical, safety, and manufacturing
- Includes Veeva Vault QMS
Hidden costs to watch for:
- Implementation fees
- Data migration
- Per-module add-ons
- Annual re-validation charges
Validation is one of the largest contributors to hidden costs; look for vendors with full GAMP 5 IQ/OQ/PQ.
| Product | Best For | Approx. Starting Price |
|---|---|---|
| MasterControl | Enterprise | Custom Quote |
| Veeva Vault QMS | Enterprise | ~$100,000+/yr (est.) |
| TrackWise | Enterprise | Custom quote |
| ETQ Reliance | Enterprise | Custom quote |
| ComplianceQuest | Mid-Market | Custom quote |
| QT9 QMS | Mid-Market | $2,200/user/yr |
| QAD EQMS | Mid-Market | Custom quote |
| Qualio | Startup / Scale-Up | $12,000/yr + $3,000/user/yr |
How to Choose QMS
When choosing pharmaceutical quality management software, ask vendors these questions before you shortlist:
Is it pre-validated?
Ask whether the QMS includes full GAMP 5 IQ/OQ/PQ, re-validation comes with updates, and whether it aligns with the FDA’s CSA framework. Some vendors include a validation fee, and others leave it to your team.
Does it support 21 CFR Part 11 natively?
Don’t just rely on the fact that audit management exists in the system. Ask to see the audit trail in the demo and how it captures what FDA inspectors expect.
Cloud or on-premises?
A cloud-based QMS offers faster deployment and cross-site accessibility. However, pharmaceutical companies with data-residency requirements may want to consider hybrid options.
Does it scale, and are workflows configurable?
Ask how the QMS manages multi-site configurations and whether CAPA, change control, and deviation workflows will match your SOPs.
What integrations does it offer?
Ask whether integrations are native, API-based, or dependent on middleware:
- ERP for material data and production orders
- LIMS for batch release and lab results
- MES for real-time shop floor data
- eTMF/regulatory systems for end-to-end connectivity
What’s the vendor’s pharma track record?
Ask for pharmaceutical customer references and whether support staff have GxP experience.
Validation Responsibility
Any computerized system in a GxP environment must be validated. Since the FDA finalized its CSA guidance in September 2025, the expectation has shifted from exhaustive CSV documentation under ISPE GAMP 5 to a risk-based approach where software validation effort is proportional to risk.
But somebody still has to do the work, and for pharmaceutical QMS software buyers, the question is: who?
| Model | What It Means |
|---|---|
| Vendor-validated platform | Vendor handles all IQ/OQ/PQ, re-validates with every update; least common approach |
| Partially validated | Vendor provides validated platform; customer completes supplier qualification, risk assessment, and/or validation summary; most common approach |
| Customer-owned | Customer owns entire validation lifecycle |
Before you sign, ask every vendor three questions:
- What does your validation package include?
- What do we need to do ourselves?
- Do we revalidate when you release a software update? Who does the work?
The answers reveal more about the true cost of ownership than any pricing sheet.
AI in Pharmaceutical QMS
Pharmaceutical QMS marketing tends to use AI liberally as a buzzword, but actual capabilities range from repackaged rules-based logic to actual workflow automation and machine learning.
- Document Change Summaries: Qualio uses AI to generate document change summaries for revised SOPs, including description, rationale, and impact
- Investigation Narratives: Veeva AI for Quality can pull from related records to create narrative investigations and CAPA summaries for human review.
- Auto-Categorization: TrackWise provides AI-assisted auto-categorization that recommends classifications for quality events and complaint intake.
- Predictive Analytics: EtQ Reliance and ComplianceQuest include predictive analytics to detect recurring issue patterns and emerging quality trends.
AI output in a GxP environment still mandates 21 CFR Part 11 sign-off and human review.
- AI in pharmaceutical QMS software isn’t a compliance shortcut; treat it more like a catalyst for continuous improvement.
- Ask vendors to demo AI with real data, and ask for customer references actually using it in production.