The Importance of Quality Control in Manufacturing
Quality control (QC) is a set of procedures a business follows to ensure that mass-produced goods meet predefined quality standards. During manufacturing, quality control involves testing units to determine the final product’s compliance with quality requirements.
In this post, learn the importance of quality control, its benefits, and its methods.
Benefits
Quality control has a direct and measurable impact on the production process. Your time and effort in screening items and monitoring variances affects the final output and consumers’ perception of your business. Consider these five benefits of QC in the manufacturing industry:
-
Cost reduction
Quality control helps reduce expenses by identifying real-time issues on the production line and averting recalls and replacements. It also distinguishes unnecessary stages in the manufacturing process that can diminish costs. -
Customer loyalty
Consumers’ perceptions of your merchandise can make or break your company. Quality control helps businesses meet customer expectations by improving the time to market, safety, and condition of your goods. Good quality offerings can lead to referrals and expand reach to new consumers. -
Efficiency
Quality control reduces downtime and streamlines workflow. QC also minimizes redundant assembly steps and helps pinpoint defective products before production is too far along. -
Safety
Quality control can diagnose potential safety concerns before goods finish assembly. When applied at a systemic level, QC prevents faulty products from injuring consumers and supports engineers in maintaining occupational health and safety standards. -
Consistent Product Quality
Ultimately, the goal of quality control in manufacturing is to provide better output. By detecting assembly line issues and poor-quality products before they reach your customers, you can improve your company’s profitability.
Quality Control vs. Assurance
Businesses follow quality management best practices to guarantee their goods meet predefined standards. These methods include quality assurance and control, which differ in the following fundamental ways:
| Quality Control | Quality Assurance |
|---|---|
| Reactive process | Proactive process |
| Product-based | Operations-based |
| Identifies defects | Prevents defects |
| Verifies quality | Manages quality |
Quality assurance finds best practices for manufacturing products, while quality control identifies issues in the manufacturing process after they occur. Both practices are critical for manufacturing plants to provide quality in the goods they produce.
Types of Methods
Quality control methods are strategic techniques that help evaluate output quality and adherence to customer requirements. Choose from the following popular approaches to bring out the best in your production process:
100% Inspection Method
This technique eliminates potential flaws by evaluating all products rather than just a sample. It’s a standard approach when assessing luxury goods, life-saving products, and raw materials.
Lean Manufacturing mMethod
This QC tool maximizes productivity and minimizes waste at every production stage. Waste includes operations or products that require time, money, and talent but that don’t present any value to consumers. The following table highlights three techniques of this method:
| Method | Explanation |
|---|---|
| 5S | A Japanese system that lays the groundwork for improvement through a well-organized workplace. 5S includes five steps: seiri (organize), seiton (orderliness), seiso (cleanliness), seiketsu (standardize), and shitsuke (discipline). |
| Kaizen | A general approach to continuous improvement based on using suggestions from all employees, small ongoing changes, and root cause analysis to resolve issues as they occur. |
| Total Productive Maintenance (TPM) | A management framework that enlists employees at every level to prevent breakdowns, accidents, production pauses, and product defects. |
Six Sigma Method
This risk management strategy minimizes deficiencies and improves the overall quality of production and business operations. It’s a statistical quality control metric that requires only 3.4 defects per one million opportunities.
Taguchi Method
This approach emphasizes product design and research over the manufacturing process. It removes the potential for variances through design work before they occur during the assembly stage.
X-Bar Chart Method
This quality control chart tracks tested samples on the x-axis and the degree to which variances are acceptable on the y-axis. Manufacturers can analyze the variance pattern to determine if defective products occur randomly or systematically.
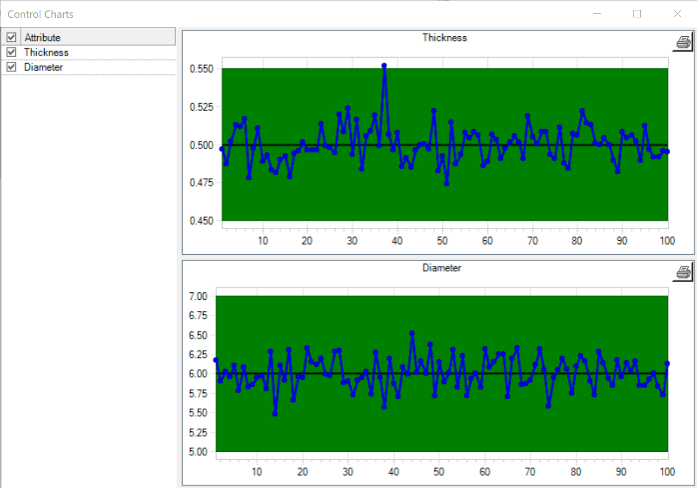
Types of Quality Audits
Manufacturing businesses conduct regular internal and external audits of their quality control measures to secure compliance in the following ways:
-
Process audits: Inspectors review work instructions, training logs, and company procedures against International Organization for Standardization (ISO) and Food and Drug Administration (FDA) regulatory requirements.
-
Product audits: Auditors inspect specific goods to ensure they meet customer requirements, performance specifications, and compliance standards.
-
System audits: Auditors review a business’s quality management system (QMS) annually against regulatory requirements, compliance standards, and contracts between the company and its consumers.
By using a quality management system, companies can define, schedule, and conduct regular audits and inspections of business operations. With a robust QMS system, manufacturers can manage workflow and set objectives on cycle times, defect rates, and scrap/waste percentages.
How to Implement Effective QA and QC
Your business’s profitability hinges on delivering high-quality products to your customer base. Auditing goods after production facilitates standards compliance and root cause analysis by tracing manufacturing issues to their origins.
To implement effective quality management, follow these steps:
-
Use proven quality control tools. Employ time-tested QC methods, like Six Sigma, to reduce variability and enhance the overall quality of your products. Constantly improve delivered products and adjust to consumers’ changing needs with agile quality management.
-
Perform checks during production. Run regular quality checks after assembly. Also, conduct audits throughout the manufacturing process. Do inspections at various production stages to identify issues as they happen while still efficiently using your time.
-
Keep track of issues. Record all quality issues to help determine solutions, whether through retooling, adjusting a process, or creating a post-production fix. Document errors to help avoid similar problems with future projects.
FAQS
What is the difference between quality control and continuous improvement?
Quality control focuses on ensuring that current deliverables meet quality standards. Continuous improvement focuses on organizational and systemic changes that indirectly enhance the development of goods.
What is the difference between quality planning and control?
Quality planning involves proactively developing a method to meet established objectives under specific operating conditions. Quality control processes are reactive and identify deficiencies within the operations prescribed by quality planning.
What is the difference between ISO 9000 and ISO 9001?
ISO 9000 is a family of internationally recognized specifications that describe universally applicable quality management principles.
ISO 9001 is a specific quality standard that organizations use, regardless of activity, to take actionable steps toward improving product quality. It ensures that every aspect of manufacturing meets globally accepted specifications.
Accredited auditors can award a certificate of compliance with ISO 9001 to compliant businesses. Learn more about the ISO 9000 standards series.
